Hard Calculations for Easy Decisions
Business purchases are driven by a sticker price. Then there’s the true, all-inclusive cost that accumulates the soft costs, the downstream costs, and even the cost of unintended consequences. Savvy decision makers attempt to consider them all, but it usually isn’t an easy exercise.
When deciding whether to invest in paper clinical outcome assessments (COAs) or electronic clinical outcomes assessments (eCOAs), as a relatively inexpensive commodity, paper may seem to be the least expensive option.
However, when all factors are considered, electronic clinical outcome assessments (eCOAs), including electronic patient reported outcomes (ePROs), eDiaries, and electronic clinician reported outcomes (eClinROs), consistently make more financial sense than paper COAs.
When is it cost effective to go electronic? How much can you save? What situations still justify a paper approach?
The Clinical ink COA Cost Calculator
To answer these questions definitively – whether for a trial or an entire development program, Clinical ink has developed a calculator that factors in and compares the hard, even if hidden, costs of each option.
The calculator considers, among other things:
Material Costs
- The paper itself
- Printing & binding of study packets
- Postage
- Archiving for an average of 15 years
Human Costs
- Preparing and mailing of study packets to sites
- The labor involved in measuring any visual analog scales (VASs) used
- Transcribing data into the database
- Data cleaning (deciphering illegible or ambiguous entries)
- Source data verification
- Data & query management
- Monitoring visits
Soft Costs of Paper COAs
- Missing and incomplete data
- Non-compliance (parking lot syndrome)
- Delayed time to market
- Regulatory risks
- Environmental impact (average tree yields only 16.67 reams of paper*)
While the true costs of paper vary due to many factors, with these inputs, the calculator can generate an accurate approximation for a per-page cost of paper COAs, that can then be multiplied by the number of patients and the number of times the COA is completed or administered to arrive at a total cost per study.
Calculated cost of paper per page X number of patients X number of times the COA is completed or administered = Cost of paper COAs in a trial.
While eCOAs can save up-to millions of dollars over paper COAs in many cases, there are a few circumstances, such as some early-phase or rare diseases trials, where having fewer participants or a limited number of COAs may warrant using paper.
While paper may be less expensive for a small phase I study, an eCOA solution may be more practical and affordable for eventual phase II and III studies. In this case, it may be wise to invest in eCOA early on to maintain consistency of data collection, and to realize the financial savings in the long run.
Calculator in Action
Running the calculator on two actual studies illustrates the magnitude of possible savings in using eCOAs over paper COAs.
Study One
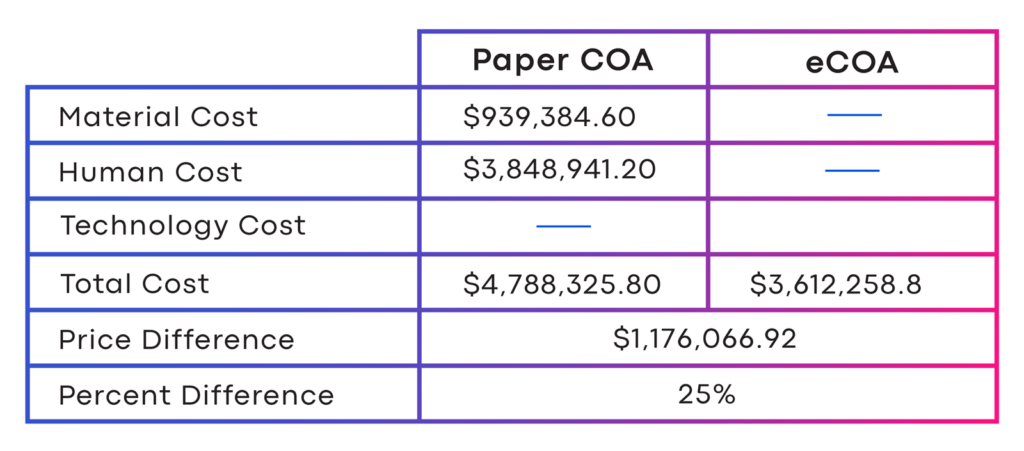
Randomized 400 subjects and required one at-home diary, eight (8) complex on-site ClinROs and four (4) on-site ePROs. In Figure 1, in total, the paper COA would cost $4.8 million. In contrast, the eCOA cost $3.6 Million.
A savings of $1.2 million or a 25 percent cost reduction.
Study Two
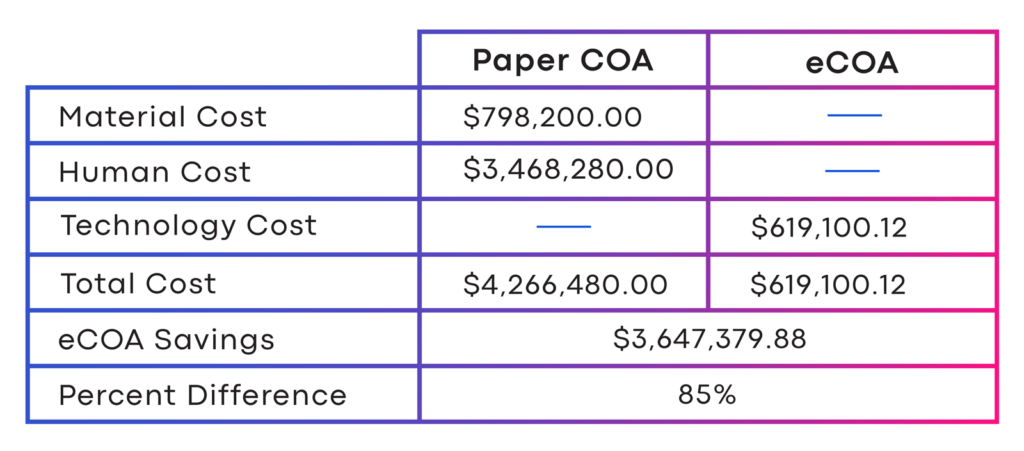
Randomized 380 subjects who were asked to complete a single at-home diary. Figure 2 shows that the cost of using paper was $4.3 million, compared to a cost of $619 thousand for the eCOA.
The electronic option represented a savings of $3.6 million, or 85 percent.
These two examples show that eCOA is typically the most economical choice, as paper options rack up substantial costs. See how this applies for your study or development program. Compare the cost of paper COA vs. eCOA so you can make a clear and compelling decision.
* http://conservatree.org/learn/EnviroIssues/TreeStats.shtml
